Injection moulding Life-saving parts: Plastic injection moulding supports intensive care medicine in times of need
Germany — Intensive care respirators manufactured for long-term treatment are particularly important during the corona crisis and were in great demand. HG Geiger Kunststofftechnik has decades of experience in the manufacture of injection moulded parts and increased production for these live-saving components on short notice.
Related Vendors
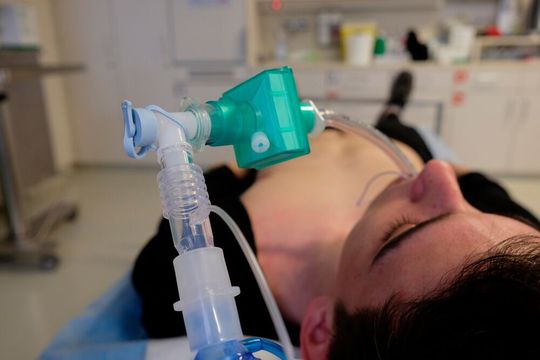
During the coronavirus pandemic, the German Government awarded contracts to various German manufacturers for the construction of a total of 20,000 respirators to ensure adequate care for all emergency patients. The high number of units and the urgency of the demand pushed both manufacturers and suppliers to their capacity limits. In order to obtain high-quality components, the medical equipment manufacturers commissioned experienced suppliers with their production. Reiter HG Geiger Kunststofftechnik in Hilpoltstein, a member of the Geiger Group, also qualified as a system supplier through 25 years of experience in the field of injection moulded parts and system assemblies for respiratory and anaesthesia equipment, as it was able to produce the required quantities within a very short time.
“With the major order from the German government for the fastest possible delivery of intensive care respirators to overcome the corona crisis, the demand for corresponding components such as adapters for respiratory gas purification also rose sharply in our company,” explains Eva Söhnlein, Managing Director of Reiter HG Geiger Kunststofftechnik. “Compared to the previous year, we recorded an increase of up to 400 percent for individual components”. The production figures were increased within a few days to cope with the demand. Reiter has expertise in various product areas, so that a wide variety of components were requested.
The portfolio of the Franconian company ranges from temperature measuring grommets and fresh gas measuring grommets, which are required in sensor technology for monitoring the patient during the anaesthesia phase, to filling and connection adapters to prevent mix-ups of anaesthetics, to complex valve assemblies for breathing gas control. “Our components are also used to record measured values and to control and regulate respiratory air flows, which must be permanently monitored, especially for corona patients,” reports Söhnlein.

Maintaining high quality standards even under time pressure
However, the production of the assemblies is a complex process. Each component requires its own injection moulding tool and the ideal production strategy must be determined at an early stage. There are numerous influencing factors here, such as the material, the area of application or the necessary steps for further processing and completion of the assembly. The complex geometries and tight manufacturing tolerances pose a particular challenge. The possible manufacturing tolerances of purchased components must also be precisely defined in advance and compensated for in the manufacturing process. In addition, depending on the area of application of the components, highly demanding plastics with specific material properties are used in production: the range extends from high-temperature plastics such as polyether ether ketone (PEEK), which exhibits physical properties such as flexural strength and tensile strength even at high temperatures of up to 260 °C, to polyphenylene sulphide (PPS), which has a high chemical resistance. Further materials are polyarylsulphone (PSU) and polyphenylsulphone (PPSU), which have a high hydrolysis stability and thus enable sterilisation of components in autoclaves.
Despite the enormous pressure caused by the high demand and shortage of existing equipment in hospitals, the products had to meet the highest quality standards.
To ensure that the components function smoothly, high quality standards also apply to all downstream processing steps such as ultrasonic and laser welding, as well as mechanical processing. The filigree nature of the individual components and the complexity of the assemblies formed from them means that manual finishing is required in many cases. This applies both to the pre-assembly of small subassemblies and the assembly of entire system modules. The high quality requirements for the products, which were already valid before the Corona crisis, also necessitate a fully documented functional test of 100 percent of the produced components using complex automatic testing machines. Both assembly and quality control take place in the clean room to ensure the technical cleanliness of the products according to DIN ISO 16232. “It is particularly important in final assembly to find the right balance between automation and manual work steps. We have combined assembly and workpiece inspection in one work step to avoid production batches of assemblies being overlooked during final acceptance. In this way, we can ensure the high quality standard of our products, for example for the manufacture of intensive care respirators,” Söhnlein sums up.
(ID:47479961)



:quality(80)/p7i.vogel.de/wcms/4f/65/4f652209fc2e425beeeb86a6553818d9/0131208808v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/2a/08/2a08f8bd2df95ca843d52cbce64bf355/0131212753v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/10/b9/10b904e03efac26f870efc9ac032bb30/0131163630v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/bb/74/bb74aefce7687b2ac87e62f6a52cf67e/0130170106v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/52/5a/525ae5678335809709ff6e1b7f900ea8/0131441227v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/e8/a7/e8a7e91741cd5be76b58e883e1d7fe27/0131384159v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/27/fe/27fe0b17ad8c67f81f869e494a88706d/0131213467v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/98/76/98766b6b22c3147596aa50c6a3a6e5f2/cjw-8143-2976x1673v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/0d/6c/0d6c5ea88cb425ab9dd066457225e61b/0131066999v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/03/41/0341f13be7d88e59075184138a9bfc44/0131010620v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/16/fd/16fd246f520f6340c1d7fe77a26f817d/0130918842v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f5/ad/f5ad2cefc6d208da995f0768238e4537/0130726865v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/9d/9e/9d9e758ce29d84f7e7e65503ca869a54/0131435800v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/40/5d/405db5313200d78f4088109353ef2935/0131353057v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/c8/a7/c8a74d047ae2600447ea8c034cc3f1aa/0131352270v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/98/a4/98a4944199d54d08edfe998f93350703/0131212199v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/a4/c2/a4c2b5c6ec1671aacae775d819ffffc7/0131434140v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/ae/38/ae38d9055a94a30815da8c0cb9d3bd22/0131212819v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/20/80/2080366dbf8cca84fe9d00659b8de85c/0131056632v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/44/b6/44b693400b1e32f96742addc69bd8d7c/0130946952v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/79/d9/79d91a893c968a8df2d8490e4f2661e9/0131465713v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/47/6b479206ca67f1323af6e58820be26e8/0131382857v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/00/9a/009ae0e133b321f8f0dabfb3fe14d2a6/0130776695v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/28/43/28438c7998aebec0c2f5fb6f1a16b387/0130764478v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d5/1d/d51def0b31e213e756bbfb9479fa9c7e/0129925621v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/f9/ca/f9ca9cace88200a5ec0946f193264963/0129209504v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/2f/61/2f61dd20be68049fa2320a2e5aa860d7/0123789183v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/c5/49/c54973f28495dada1c7605f913717341/0121533734v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/76/aa/76aa92f11f136c10e16cc77a89a5f583/0131009272v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/53/c7/53c73ae0a157d4c103e1964e77c1f425/0130023300v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/68/ab/68abf66929cb9f2dcb823a12e90d1d21/0129922525v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/46/85/46854ab7a2679db67354f556f2bc1db9/0129539660v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/60/45/6045cb15d71397154c6cee56d5d95ac7/0131063997v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/4c/dd/4cdd1643347d5f6f72743402d36e93f4/0130628083v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/51/3e/513e2d937e96401a4d1ff8db1481cce7/0130242596v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/f8/8f/f88f47ba8e4ef50152497010c25dda11/0130168360v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/be/4d/be4da3d2f0d802d94127979ea4af68b4/0130748997v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/66/52/6652eb3a653a0908d5b03fb638f16260/0129646074v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/9e/bd/9ebd826ceed28c644a9205bdbf82beb4/0131007997v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/d5/e6/d5e68c74fabff18ef737ee767b7b359f/0130415707v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/6b/9c/6b9cab1e262f77caed8179ed3dc29807/0129104421v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/52/97/52978cbd7dfc421404314e34b8348a8a/0131353724v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/4d/fa/4dfa0b367fb9c8da795c7fc29dc0815c/0131301905v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/ce/80/ce8008acc51e05d5c33a5890d07e239d/0131117485v1.jpeg)
:quality(80)/p7i.vogel.de/wcms/e7/74/e774210496e99be6481defcde77bab19/0131116903v1.jpeg)
:fill(fff,0)/p7i.vogel.de/companies/66/0e/660e93698db24/hasco-logo-or-b50.jpeg)
:fill(fff,0)/images.vogel.de/vogelonline/companyimg/26200/26226/65.jpg)
:quality(80)/p7i.vogel.de/wcms/10/f8/10f8a23728a41e4dc857acd16c8eb61c/0125079533v2.jpeg)
:quality(80)/p7i.vogel.de/wcms/19/46/1946128e219613a59adb20ce3dbf1386/0127812088v1.jpeg)